19 de August de 2020
Does Time of Day Affect Sports Performance?
Everyone knows that performance varies, even on a day-to-day basis. This variation is not trivial and you don´t need a fancy and complex methodology to detect it. We´ve all been there, running our best time, being the man of the match or nailing a PB one day and struggling with light weights, losing a race with a slower partner or making stupid mistakes in an easy competition, match or whatever your sports call it.
It seems that the time of day at which a task is done can be one of the many factors that can affect performance
I´m a former taekwondo athlete with a decent career with ups and downs as every other athlete in the world. But one of the most frustrating things I remember back on the day I was competing at the highest level was those shitty inexplicable days where things turned out to be just harder than usual. Both physically, and mentally things simply felt off and easy tasks that were fully automated and done a million times didn’t felt easy at all.
Of course, performance is a complex concept and its variation can not be explained by a single phenomenon. However, it seems that the time of day at which a task is done can be one of the many factors that can affect performance. Various psychological and physiological functions (if not all), have been shown to undergo changes relative to the time of day1. These variations are known as circadian or diurnal rhythms 2. The majority of components of sports performance, e.g. flexibility, muscle strength, explosiveness, vary with time of day in a rhythmic manner 1,3.
But, Is there an optimal time of day to train or perform?; Are the time of day fluctuation of performance relevant? Can our habits affect our body rhythms and thus performance?
Circadian Rythm And Biological Clocks
The term Circadian comes from the Latin “circa” (around) and “dies” (day) and refers to the fact that a great variety of biological processes “oscillates” in a cycle of approximately 24 hours. These oscillations allow the human body to assign functions according to the time of day, prioritizing certain tasks according to sleep-wake cycles (eg: repair tissue during sleep, metabolic processes during the day/activity)4.
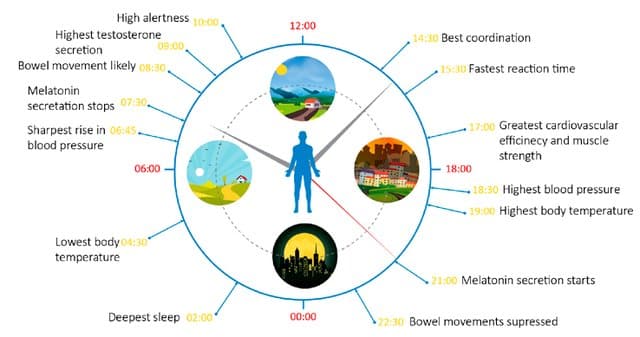 Figure 1 Human circadian rhythm 5
Figure 1 Human circadian rhythm 5
Circadian rhythms are the biological expression of time and a way to anticipate environmental events. So, the proper functioning of these oscillations is crucial for health and performance. In each one of our cells, there is a molecular mechanism in charge of functioning as a Clock which necessarily has to be synchronized with the environmental information (cues) and that governs every aspect of biology, from body temperature, hormonal regulation and activity patterns, sleep-wake cycle and even cellular processes6–8.
The internal generator of rhythms is located in the brain, specifically in an area called the Suprachiasmatic Nucleus (hypothalamus). The so-called “Central Clock”, about 20,000 nerve cells are in charge of regulating and synchronizing all peripheral cell clocks in a similar way to that of an orchestra director conducts a symphony9.
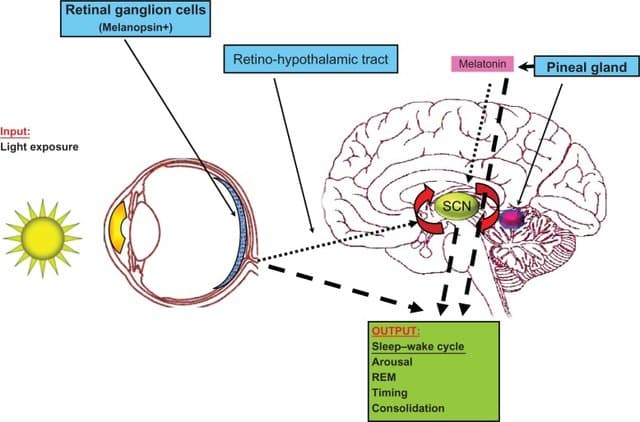 Figure 2 Functioning of the master clock in the suprachiasmatic nucleus (SCN)10
Figure 2 Functioning of the master clock in the suprachiasmatic nucleus (SCN)10
This central clock works in a “similar” way to a solar clock since the main source of information on which it is based to “know” the time is Light during the day and darkness at night, which generates the so-called diurnal cycle or light-dark cycle11. The molecular mechanism underlying circadian rhythms is a gene-regulatory network composed of transcriptional-translational feedback loops referred to as the “core clock” and it´s present in every single cell12,13. And because free-running clocks cycles without any external information appear to be longer than 24hs (~25hs) a key aspect of circadian physiology is properly entraining to a 24 hs cycle with external signals or natural time synchronisers (referred to as the German term “zeitgeber”) that have the ability to “rewind” the biological clock.
It governs every aspect of biology, from body temperature, hormonal regulation and activity patterns, sleep-wake cycle and even cellular processes
Light is the main zeitgeber but not the only one, the timing of food intake (Chrononutrition), physical activity and some social and psychological factors are also considered as zeitgebers and through the main biological clock, affect the peripheral oscillators that regulate many important physiological functions of metabolism12,13.
The vast majority of hormones (and indeed cellular processes) exhibit a diurnal rhythm. Also, the sensitivities of different cell types to different hormones also exhibit oscillatory behavior (Chronopharmacology studies the impact of different medications on different pathologies depending on the time of day when it is administered). This has an impact on every cell and tissue in the body; the immune system has circadian oscillations, making us more or less vulnerable to infections and allergies14, insulin secretion as well, causing the pancreatic response to a carbohydrate-rich meal to be not the same depending on the time of day15, strength appears to peak in the later afternoon versus morning 1 while oxidative capacity increases in the late evening 16.
Moreover, circadian rhythms, among other factors, have also been shown to, not only regulate key physiological processes involved in athletic performance 3,17–21 but also disruption in circadian rhythms can have a negative impact in both physical and cognitive performance1,4,22.
Time of day and performance.
Generally speaking, it appears that personal records tent to occur in the evening across different sports23–28 and it is likely because of the behavior of the body temperature which tends to reach its lowest around 04:30h and increases gradually till its peak at 18:00h. Raising body temperature can facilitate the actin and myosin mechanics in the musculoskeletal structures, thus, improving muscle contraction properties and in result physical performance29. Moreover, higher body temperature shifts metabolic fuel utilization towards carbohydrates which may improve performance as well30. Moreover, the increased temperature in the afternoon strongly correlates with increased coordination, the highest reaction time, increasing muscle strength and cardiovascular efficiency 1,31. In addition, anaerobic power output and flexibility are also the highest in the late afternoon 32.
Raising body temperature can improve muscle contraction properties and in result physical performance
Some authors emphasize that temperature is not the only factor affecting performance variations during the day 33. State of vigilance, alertness, and mood levels (which are highest in the morning) can affect athletic performance 34. Even though simple mental tasks such as reaction time may depend on body temperature (higher temperatures enhanced neural activity; for every 1°C rise in body temperature, nerve conduction velocity increases by 2.4 m1sec1), other more complex mental actions like decision making, short term memory, concentration and reasoning peaks around noon30. This may be because there is an inverse relationship between speed and accuracy. Additionally, some of those tasks require a low arousal level.
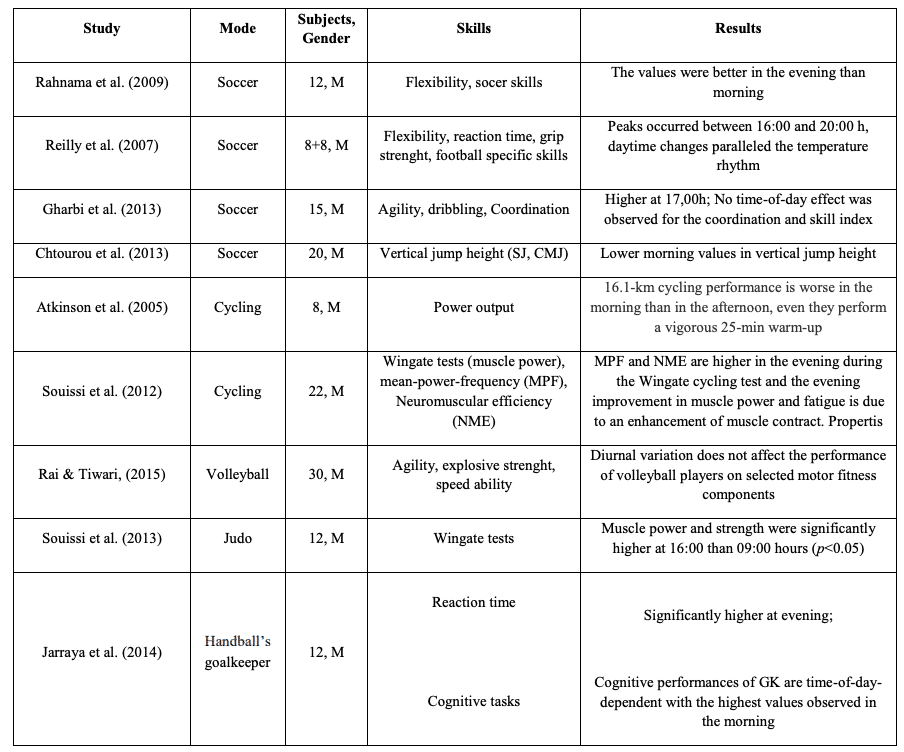 Figure 3 Summary of some findings in impact of circadian rhythm on performance 30
Figure 3 Summary of some findings in impact of circadian rhythm on performance 30
State of vigilance, alertness, and mood levels (which are highest in the morning) can affect athletic performance
Chronotypes
One of the key factors that can affect, not only how, but also when and how much impact daily fluctuations have on physiological aspects, are chronotypes 21.
The fact that the SCN does not always keep to a cycle of exactly 24 hours explains another phenomenon: Circadian chronotype, which is typically defined by a person’s preferred time to conduct activities, and/or the timing of sleep, assessed by questionnaires35 or Munich chronotype 36 questionnaires which has a well-documented genetic 37–39 and physiological26,40–42 differences that result in disparities between their biological clocks and how they entrain to exogenous cues, such as the environmental light/dark cycle and social factors. Some people can be characterized as “owls” and some people as “larks”. This distinction is a colloquial way of expressing that there are some subgroups of people who would prefer to wake later in the day and go to bed late (the owls) and there are others who wake early and go to bed early (the larks)35,36.
It appears that athletes with extreme morning or evening chronotypes tend to perform better near their own circadian peak in endurance events
At the individual level, peak performance is also influenced by circadian phenotypes, including an individual’s diurnal preference for morning or evening activity patterns and chronotype 17,39. So, it appears that athletes with extreme morning or evening chronotypes tend to perform better near their own circadian peak in endurance events 24,43, and also in strength-related tasks27,28,44. Moreover, these difference between chronotypes may be strong enough to shape the distribution of circadian phenotypes in some endurance sports that regularly compete in the morning (it seem that morning-types are more prevalent among elite runners, cyclists, and triathletes42,45).
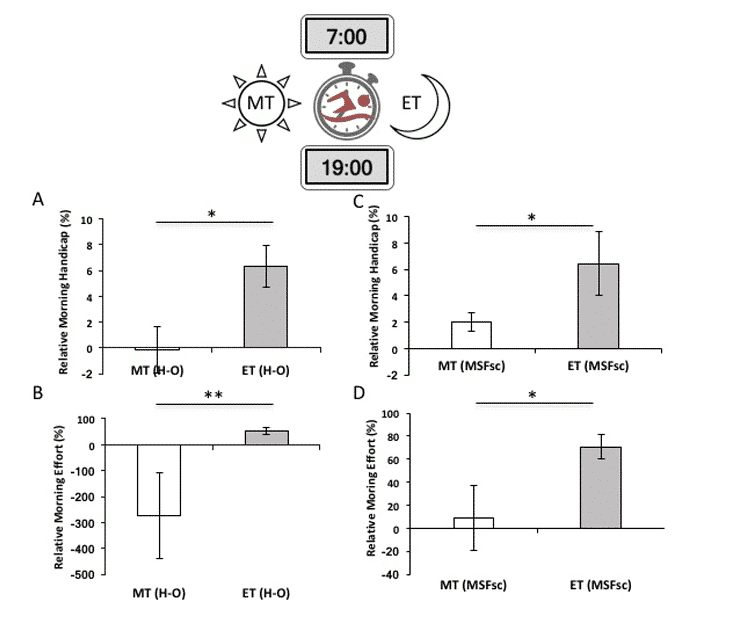 Figure 4 diffence between Morning Type (MT) vs Evening Type (ET) in performance and effort measurements for circadian phenotypes measured by diurnal preference and chronotypes. Evening-types, have a significantly greater morning handicap than morning-types, swimming 6% slower in morning (07h00) than evening (19h00); Both MT and ET participants exert more effort at off-peak times and there is a significant relative percent difference in diurnal effort between MT and ET25 .
Figure 4 diffence between Morning Type (MT) vs Evening Type (ET) in performance and effort measurements for circadian phenotypes measured by diurnal preference and chronotypes. Evening-types, have a significantly greater morning handicap than morning-types, swimming 6% slower in morning (07h00) than evening (19h00); Both MT and ET participants exert more effort at off-peak times and there is a significant relative percent difference in diurnal effort between MT and ET25 .
Moreover, chronotype may influence the psychophysiological responses to exertion, including rate of perceived exertion (RPE), heart rate variability (HRV) and mood 23,46–48. Peak performance appears to have a strong correlation with peak body temperature, which tends to occur around early evening. However, the strongest biological markers of the expression of the circadian rhythms, such as melatonin, cortisol and even body temperature differ between morning and evening chronotypes40,49. Thus, individual physiological differences may affect performance, especially on extreme chronotypes. Moreover, it appears that the major predictor of peak performance times is the time since entrained awakening, rather than the time of day, as well as significant individual performance variations, can be as large as 26% in the course of a day21.
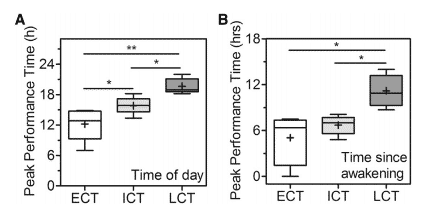 Figure 5 . Peak Performance Times as Functions of Time of Day expressed as real-time (A) and Time since Entrained Awakening expressed as relative time since entrained (B) between different chronotypes (ECT=Early Chronotype; ICT= Intermediate Chronotype; LCT= Late Chronotype).
Figure 5 . Peak Performance Times as Functions of Time of Day expressed as real-time (A) and Time since Entrained Awakening expressed as relative time since entrained (B) between different chronotypes (ECT=Early Chronotype; ICT= Intermediate Chronotype; LCT= Late Chronotype).
Circadian timing has also been reported to affect exercise outcomes at the molecular level. Two of the most researched cellular signalling pathways involved in exercise mediated adaptations, mechanistic target of rapamycin complex 1 (mTORC1), which is mainly involved in the protein synthesis response and peroxisome proliferator-activated receptor-gamma coactivator 1 (PGC1), which is related to mitochondria biogenesis, have been shown to be strongly influenced by the molecular clock and both exhibits time dependant activity50,51.
Finally, heterogeneity or variability in exercise adaptation responses (whether it be resistance or endurance training) has led to the development of terms such as “non,” “low,” “moderate,” “high,” and “extreme” responders to describe the divergent magnitude in the response of a particular individual to a given exercise stimulus52
In addition, it has been suggested that the magnitude of the adaptative response following resistance or endurance training might be due to the circadian clocks at a cellular level52.
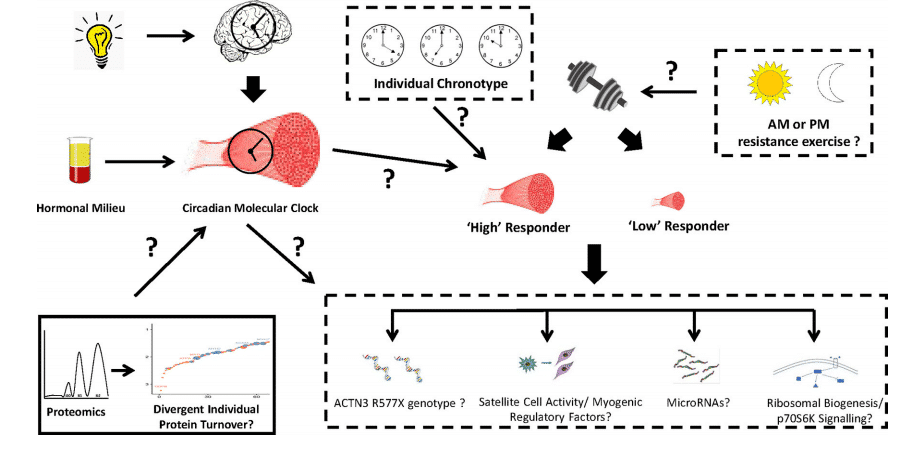 Figure 6 Hypothetical link between the skeletal muscle molecular clock and resistance exercise adaptation responses52.
Figure 6 Hypothetical link between the skeletal muscle molecular clock and resistance exercise adaptation responses52.
Practical Applications
First of all, is crucial to acknowledge that circadian rhythms are not a fixed, rigid variable and they can be “manipulated”. This means that if you regularly train at the same time of day you will probably get used to that schedule and if for some reason you need to change your training time, let’s say from 19:00h to 9:00, you will eventually adapt. Furthermore, exercise is a powerful peripheral circadian zeitgeber which means that it can manipulate biological clocks. In fact, as we age our rhythms become less robust and exercise appears to reverse it53.
If you regularly train at the same time of day you will probably get used to that schedule and if for some reason you need to change your training time
The optimal time for performance will be dependent on the type of activities required in the sport and their relative importance to the overall performance. this means that every athlete should analyze the pros and cons of manipulating the time of training sessions and decide accordingly.54 On the other hand, some schedules are not possible to change. For example, a football team can not organize training sessions based on chronotypes (at least not now). Moreover, competition time is also fixed and if you know it in advance it may be beneficial to manipulate your training schedule in order to adapt.
Sun exposure is probably the most important and effective tool to strengthen the circadian cycle
Caution must be taken from this information. Most people have intermediate chronotype and it would probably be fine regardless of his training time (of course, taking into account reasonable training hours). However, some extreme chronotypes may be more sensitive to training at different hours but, it´s important to notice that even the most extreme night owls won’t benefit from training at 3 am ..in fact, there is a thin line between extreme chronotypes and circadian desynchronization which is pathological and need medical intervention55–57. Even in anytime of the day you can use a Vitruve encoder to mesure your velocity based training sessions.
Finally, and more importantly, everyone could benefit from taking care of your biological rhythms and avoiding circadian misalignments or arrhythmias regardless of your training level, schedule or chronotype. Sun exposure is probably the most important and effective tool to strengthen the circadian cycle beyond the health benefits of the sun per se, and with a few minutes after waking up (about 30 minutes) it would be more than enough, the trick is to do it at the same time always58,59.
Furthermore, popular gadgets such as cell phones and tablets are in very common use mainly at night to watch movies, social media or just leisure. Every source of artificial light has been shown to be a powerful SUPPRESSOR of melatonin production and a circadian disruptor60–63. Avoiding artificial light, especially at night, is essential to ensure restful sleep and avoid circadian insults. Ideally, when the sun starts to fall, our exposure to artificial light should decrease. I know this is not an easy task but with the help of science, it can be done. Researchers have shown that the use of blue light blocking glasses can minimize the harmful impact of artificial light64–67. So, using blue-blocking glasses as the sun goes down can be a fantastic tool to avoid sleep issues, circadian perturbation and the negative impact in both health and performance.
Bibliography
- Atkinson, G. & Reilly, T. Circadian Variation in Sports Performance. Sports Medicine 21, 292–312 (1996).
- Daido, H. Why Circadian Rhythms are Circadian: Competitive Population Dynamics of Biological Oscillators. Physical Review Letters 87, (2001).
- Chtourou, H. & Souissi, N. The Effect of Training at a Specific Time of Day. Journal of Strength and Conditioning Research 26, 1984–2005 (2012).
- Manfredini, R., Manfredini, F., Fersini, C. & Conconi, F. Circadian rhythms, athletic performance, and jet lag. British Journal of Sports Medicine 32, 101–106 (1998).
- Bjelajac, D. & Đerčan, B. Artificial light at night as an unrecognized threat to protected areas of Autonomous Province of Vojvodina (North Serbia). Zbornik radova Departmana za geografiju, turizam i hotelijerstvo 46–56 (2019). doi:10.5937/zbdght1901046b
- Takahashi, J. S. Molecular components of the circadian clock in mammals. Diabetes, Obesity and Metabolism 17, 6–11 (2015).
- Takahashi, J. S. Molecular genetics of circadian clocks in mammals. GBM Annual Spring meeting Mosbach 2003 2003, (2003).
- Ko, C. H. & Takahashi, J. S. Molecular components of the mammalian circadian clock. Human Molecular Genetics 15, R271–R277 (2006).
- Welsh, D. K., Takahashi, J. S. & Kay, S. A. Suprachiasmatic Nucleus: Cell Autonomy and Network Properties. Annual Review of Physiology 72, 551–577 (2010).
- Avidan, A. & Colwell, C. Jet lag syndrome: circadian organization, pathophysiology, and management strategies. Nature and Science of Sleep 187 (2010). doi:10.2147/nss.s6683
- Bhadra, U., Thakkar, N., Das, P. & Pal Bhadra, M. Evolution of circadian rhythms: from bacteria to human. Sleep Medicine 35, 49–61 (2017).
- Harfmann, B. D., Schroder, E. A. & Esser, K. A. Circadian Rhythms, the Molecular Clock, and Skeletal Muscle. Journal of Biological Rhythms 30, 84–94 (2014).
- Schroder, E. A. & Esser, K. A. Circadian Rhythms, Skeletal Muscle Molecular Clocks, and Exercise. Exercise and Sport Sciences Reviews 41, 224–229 (2013).
- Clockwork allergy: How the circadian clock underpins allergic reactions. Journal of Allergy and Clinical Immunology 142, 1032 (2018).
- Basse, A. L. et al. Skeletal Muscle Insulin Sensitivity Show Circadian Rhythmicity Which Is Independent of Exercise Training Status. Frontiers in Physiology 9, (2018).
- Van Moorsel, D. et al. Demonstration of a day-night rhythm in human skeletal muscle oxidative capacity. Molecular Metabolism 5, 635–645 (2016).
- Souissi, N., Gauthier, A., Sesboüé, B., Larue, J. & Davenne, D. Effects of regular training at the same time of day on diurnal fluctuations in muscular performance. Journal of Sports Sciences 20, 929–937 (2002).
- Chtourou, H. et al. The Effect of Strength Training at the Same Time of the Day on the Diurnal Fluctuations of Muscular Anaerobic Performances. Journal of Strength and Conditioning Research 26, 217–225 (2012).
- Callard, D., Davenne, D., Gauthier, A., Lagarde, D. & Van Hoecke, J. CIRCADIAN RHYTHMS IN HUMAN MUSCULAR EFFICIENCY: CONTINUOUS PHYSICAL EXERCISE VERSUS CONTINUOUS REST. A CROSSOVER STUDY. Chronobiology International 17, 693–704 (2000).
- Hower, I. M., Harper, S. A. & Buford, T. W. Circadian Rhythms, Exercise, and Cardiovascular Health. Journal of Circadian Rhythms 16, (2018).
- Facer-Childs, E. & Brandstaetter, R. The Impact of Circadian Phenotype and Time since Awakening on Diurnal Performance in Athletes. Current Biology 25, 518–522 (2015).
- Wolff, C. A. & Esser, K. A. Exercise timing and circadian rhythms. Current Opinion in Physiology 10, 64–69 (2019).
- Kunorozva, L., Roden, L. C. & Rae, D. E. Perception of effort in morning-type cyclists is lower when exercising in the morning. Journal of Sports Sciences 32, 917–925 (2014).
- Kline, C. E. et al. Circadian Variation in Swim Performance. Medicine & Science in Sports & Exercise 38, S226–S227 (2006).
- Anderson, A. et al. Circadian Effects on Performance and Effort in Collegiate Swimmers. Journal of Circadian Rhythms 16, (2018).
- Brown, F. M., Neft, E. E. & LaJambe, C. M. Collegiate Rowing Crew Performance Varies by Morningness-Eveningness. Journal of Strength and Conditioning Research 22, 1894–1900 (2008).
- Küüsmaa, M. et al. Effects of morning versus evening combined strength and endurance training on physical performance, muscle hypertrophy, and serum hormone concentrations. Applied Physiology, Nutrition, and Metabolism 41, 1285–1294 (2016).
- Malhotra, D., Narula, R., Zutshi, K., Kapoor, G. & Aslam, B. Effect of Time of Day and Concentric or Eccentric Strength Training on Muscle Strength. Indian Journal of Physiotherapy and Occupational Therapy – An International Journal 8, 134 (2014).
- Starkie, R. L., Hargreaves, M., Lambert, D. L., Proietto, J. & Febbraio, M. A. Effect of Temperature on Muscle Metabolism During Submaximal Exercise in Humans. Experimental Physiology 84, 775–784 (1999).
- Vitosevic, B. The circadian clock and human athletic performance. The University Thought – Publication in Natural Sciences 7, 1–7 (2017).
- Shibata, S. & Tahara, Y. Circadian rhythm and exercise. The Journal of Physical Fitness and Sports Medicine 3, 65–72 (2014).
- RACINAIS, S., BLONC, S., JONVILLE, S. & HUE, O. Time of Day Influences the Environmental Effects on Muscle Force and Contractility. Medicine & Science in Sports & Exercise 37, 256–261 (2005).
- Robinson, W. R. et al. Does Lowering Evening Rectal Temperature to Morning Levels Offset the Diurnal Variation in Muscle Force Production? Chronobiology International 30, 998–1010 (2013).
- Atkinson, G. & Speirs, L. Diurnal Variation in Tennis Service. Perceptual and Motor Skills 86, 1335–1338 (1998).
- Horne, J. A. & Östberg, O. Individual differences in human circadian rhythms. Biological Psychology 5, 179–190 (1977).
- Zavada, A., Gordijn, M. C. M., Beersma, D. G. M., Daan, S. & Roenneberg, T. Comparison of the Munich Chronotype Questionnaire with the Horne‐Östberg’s Morningness‐Eveningness score. Chronobiology International 22, 267–278 (2005).
- Dijk, D.-J. & Archer, S. N. PERIOD3, circadian phenotypes, and sleep homeostasis. Sleep Medicine Reviews 14, 151–160 (2010).
- Katzenberg, D. et al. A CLOCK Polymorphism Associated with Human Diurnal Preference. Sleep 21, 569–576 (1998).
- Carpen, J. D., von Schantz, M., Smits, M., Skene, D. J. & Archer, S. N. A silent polymorphism in the PER1 gene associates with extreme diurnal preference in humans. Journal of Human Genetics 51, 1122–1125 (2006).
- Bailey, S. L. & Heitkemper, M. M. CIRCADIAN RHYTHMICITY OF CORTISOL AND BODY TEMPERATURE: MORNINGNESS-EVENINGNESS EFFECTS. Chronobiology International 18, 249–261 (2001).
- Nováková, M., Sládek, M. & Sumová, A. Human Chronotype Is Determined in Bodily Cells Under Real-Life Conditions. Chronobiology International 30, 607–617 (2013).
- Lack. Chronotype differences in circadian rhythms of temperature, melatonin, and sleepiness as measured in a modified constant routine protocol. Nature and Science of Sleep 1 (2009). doi:10.2147/nss.s6234
- Bougard, C., Bessot, N., Moussay, S., Sesboüé, B. & Gauthier, A. Effects of Waking Time and Breakfast Intake Prior to Evaluation of Physical Performance in the Early Morning. Chronobiology International 26, 307–323 (2009).
- Küüsmaa, M., Sedliak, M. & Häkkinen, K. Effects of time-of-day on neuromuscular function in untrained men: Specific responses of high morning performers and high evening performers. Chronobiology International 32, 1115–1124 (2015).
- Kunorozva, L., Stephenson, K. J., Rae, D. E. & Roden, L. C. Chronotype andPERIOD3Variable Number Tandem Repeat Polymorphism in Individual Sports Athletes. Chronobiology International 29, 1004–1010 (2012).
- Rae, D. E., Stephenson, K. J. & Roden, L. C. Factors to consider when assessing diurnal variation in sports performance: the influence of chronotype and habitual training time-of-day. European Journal of Applied Physiology 115, 1339–1349 (2015).
- Vitale, J. A. & Weydahl, A. Chronotype, Physical Activity, and Sport Performance: A Systematic Review. Sports Medicine 47, 1859–1868 (2017).
- Vitale, J. A., La Torre, A., Baldassarre, R., Piacentini, M. F. & Bonato, M. Ratings of Perceived Exertion and Self-reported Mood State in Response to High Intensity Interval Training. A Crossover Study on the Effect of Chronotype. Frontiers in Psychology 8, (2017).
- Baehr, E. K., Revelle, W. & Eastman, C. I. Individual differences in the phase and amplitude of the human circadian temperature rhythm: with an emphasis on morningness-eveningness. Journal of Sleep Research 9, 117–127 (2000).
- Um, J.-H. et al. AMPK Regulates Circadian Rhythms in a Tissue- and Isoform-Specific Manner. PLoS ONE 6, e18450 (2011).
- Ramanathan, C. et al. mTOR signaling regulates central and peripheral circadian clock function. PLOS Genetics 14, e1007369 (2018).
- Camera, D. M. Anabolic Heterogeneity Following Resistance Training: A Role for Circadian Rhythm? Frontiers in Physiology 9, (2018).
- Nakamura, T. J. et al. Age-Related Decline in Circadian Output. Journal of Neuroscience 31, 10201–10205 (2011).
- Drust, B., Waterhouse, J., Atkinson, G., Edwards, B. & Reilly, T. Circadian Rhythms in Sports Performance—an Update. Chronobiology International 22, 21–44 (2005).
- Lucassen, E. A. et al. Environmental 24-hr Cycles Are Essential for Health. Current Biology 26, 1843–1853 (2016).
- Granada, A. E., Cambras, T., Díez-Noguera, A. & Herzel, H. Circadian desynchronization. Interface Focus 1, 153–166 (2010).
- Arble, D. M., Ramsey, K. M., Bass, J. & Turek, F. W. Circadian disruption and metabolic disease: Findings from animal models. Best Practice & Research Clinical Endocrinology & Metabolism 24, 785–800 (2010).
- Van der Rhee, H. J., de Vries, E. & Coebergh, J. W. Regular sun exposure benefits health. Medical Hypotheses 97, 34–37 (2016).
- Bonmati-Carrion, M. et al. Protecting the Melatonin Rhythm through Circadian Healthy Light Exposure. International Journal of Molecular Sciences 15, 23448–23500 (2014).
- Gringras, P., Middleton, B., Skene, D. J. & Revell, V. L. Bigger, Brighter, Bluer-Better? Current Light-Emitting Devices – Adverse Sleep Properties and Preventative Strategies. Frontiers in Public Health 3, (2015).
- Gooley, J. J. et al. Exposure to Room Light before Bedtime Suppresses Melatonin Onset and Shortens Melatonin Duration in Humans. Endocrine Reviews 32, 155–156 (2011).
- Cheung, C. H. M., Bedford, R., Saez De Urabain, I. R., Karmiloff-Smith, A. & Smith, T. J. Daily touchscreen use in infants and toddlers is associated with reduced sleep and delayed sleep onset. Scientific Reports 7, (2017).
- Brainard, G. C. et al. Dose-response relationship between light irradiance and the suppression of plasma melatonin in human volunteers. Brain Research 454, 212–218 (1988).
- Janků, K., Šmotek, M., Fárková, E., Miletínová, E. & Kopřivová, J. Blue light blocking glasses and CBT-I: effect on subjective and objective sleep quality. Sleep Medicine 64, S174 (2019).
- Esaki, Y. et al. Wearing blue light-blocking glasses in the evening advances circadian rhythms in the patients with delayed sleep phase disorder: An open-label trial. Chronobiology International 33, 1037–1044 (2016).
- Yu, Y. G. & Choi, E. J. A Study on Blue Light Blocking Performance and Prescription for Blue Light Blocking Lens. Journal of Korean Ophthalmic Optics Society 18, 297–304 (2013).
- Lockley, S. W. Safety considerations for the use of blue-light blocking glasses in shift-workers. Journal of Pineal Research 42, 210–211 (2007).

