2 de December de 2020
Strength and science Velocity Based Training
Unique properties and adaptations followed by eccentric training. Part II
In the previous article we talked about the scientific background of eccentric training and its physiological characteristics. In this article we will take a look at the main physiological adaptations that occur after eccentric training. Contraction type appears to mediate a region-specific hypertrophy; eccentric training tends to induce greater increases in distal muscle size, while mid-muscle hypertrophy occurs to a greater extent following concentric training35,36. Additionally, fiber type composition may be uniquely influenced by eccentric training, and either increase or maintenance, of IIx(IIb) fibers, has been found compared with concentric training27,36,37.
3.1.1. DOMS and EIMD
One of the negative aspects of eccentric loading is that it induces greater muscle damage and negative acute functional consequences than other types of exercise38. The combination of high force and reduced fiber recruitment causes high mechanical stress on the striated muscle that may lead to focal microlesions of the muscle fibers39.
Several studies described molecular changes within the sarcomere and myofibers following intense and/or unaccustomed eccentric exercise 40,41. The sarcomeric disorganization has been associated with disruptions to the sarcolemma and the extracellular matrix, swelling of mitochondria, and fragmentation of the sarcoplasmic reticulum42,43.
The cellular disruption of the muscle fiber triggers an inflammatory response due to the secretion of pro-inflammatory cytokines which leads to an immune response mediated by the infiltration of neutrophils and macrophages44. The inflammation would ensure the removal of tissue debris from the injured area and promote muscle repair by activating satellite cells in a process called chemotaxis45–47.
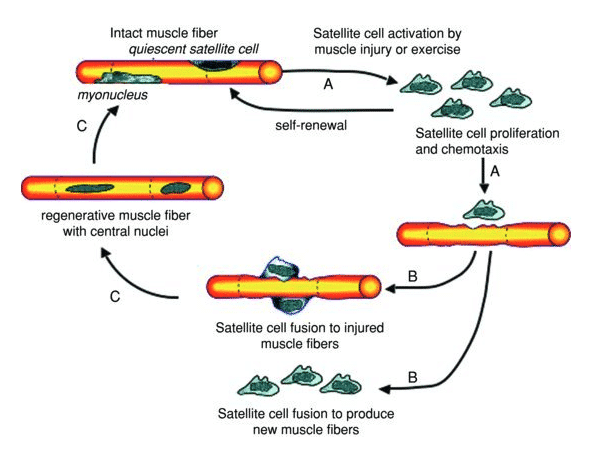
Figure 6 Schematic representation of the cycle of satellite cell activation, proliferation, and differentiation following muscle injury44.
Exercise-induced muscle damage (EIMD) is observed immediately after exercise, can gradually extend to a larger number of muscle fibers and appears to exacerbate 2–3 days post-exercise48. Furthermore, multiple factors such as muscle architecture, muscle typology, individual fitness, age, sex, and genetic variability may contribute to the wide inter-subject variability in the response to eccentric exercise24,41,49. The EIMD manifests itself by a wide range of clinical symptoms including delayed-onset muscle soreness (DOMS), stiffness, swelling and various functional deficits such as a loss in force-generating capacity or decreased proprioceptive function41.
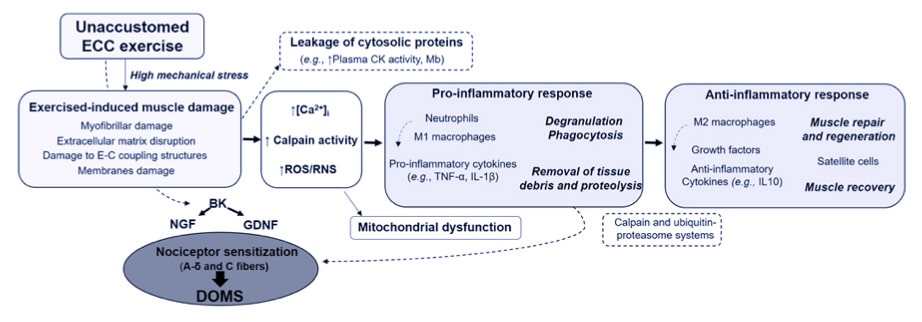
Figure 7 Summary of the main specific features of eccentric contraction, its multi-target beneficial effects and potential risks associated with unaccustomed and/or maximal eccentric exercise41.
3.1.2. Repeated Bout Effect
On the other hand, there is conclusive evidence that the first bout of eccentric exercise confers protection against EIMD following a subsequent bout of the similar exercise. This process is best known as “the repeated-bout effect” (RBE) and is characterized by a lesser inflammatory and damaging response, attenuated DOMS, less muscle swelling, faster recovery of muscle strength and range of motion50,51. Even though a single bout may confer protective effects52; the magnitude of the protection appears to increase after several sessions53 and it seems to persist for several weeks or even month although the effect diminishes over time54,55. Furthermore, there is no relationship between the magnitude of the damage and the protective effect since it has been demonstrated that repeating bouts of “non-damaging” eccentric exercise can provide strong protective adaptations against subsequent bouts of maximal eccentric exercise56.
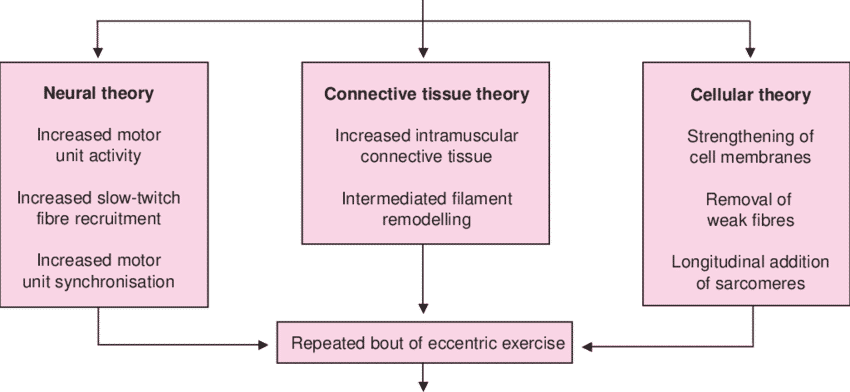
Figure 8 Potential mechanisms which may explain the repeated bout effect57.
Several theories have been proposed to explain the RBE, and it seems that there is a multifactorial origin. Potential adaptations have been categorized as neural58, mechanical59 and cellular51. However, although many studies have attempted to elucidate the mechanisms behind the RBE, a unified theory is not yet available57.
3.1.3. Muscle architecture.
We´ve already pointed out that muscle growth is achieved with both CON and ECC modalities. However, the mechanisms of structural remodelling seem to be contraction-specific60. ECC results in a markedly greater increase in fascicle length while CON promotes greater changes in pennation angle (PA), likely reflecting the differential addition of sarcomeres either in series or in parallel, respectively36,61. Thus, changes in muscle architecture could have an impact on the functional properties of muscles62. Moreover, it has been suggested that contractile material placed in series can have a “protective” impact after eccentric EIMD63, also because of the increase of maximal force produced at longer muscle lengths64. Furthermore, It has been also shown that ECC loading may positively impact tendon force65 and extracellular matrix remodelling66.
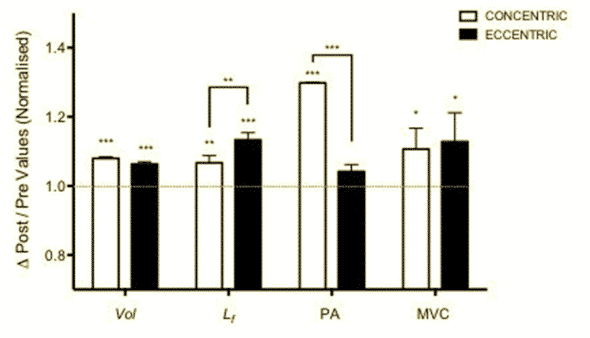
Figure 9 Contraction-dependent muscle growth in response to eccentric and concentric resistive training. Fascicle length (Lf) shows greater improvement after ECC training while Pennation angle (PA) increases after CON training14 .
3.1.3.1. Length-Tension Relationship
The length-tension relationship plays a very important role in the function of skeletal muscle. The magnitude of force a muscle can generate depends on its length, velocity and stimulation67. ECC contraction has also important implications in rehabilitation settings because it can influence the shift of the optimum length-tension relationship, with notable repercussions on performance and injury prevention67. In fact, the addition of sarcomeres in series appears to directly impact the maximum velocity of shortening of muscle fibers. Thus, eccentric contractions, while favoring the increase in Lf without presenting significant changes in PA, may have a profound influence on muscle performance68
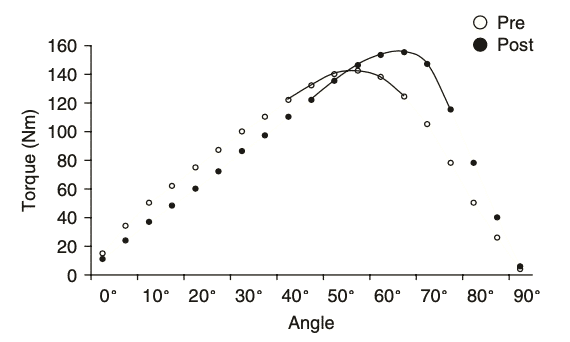
Figure 10 Schematic representation of a shift in the optimum length-tension relationship67
Neural Adaptations
It seems that eccentric contractions exhibit unique neural characteristics compared with concentric and isometric contractions under both maximal and submaximal loads indicating that the nervous system employs unique activation strategies during eccentric 10,69.
When compared with concentric or isometric contractions, eccentric actions seem to have lesser recruitment and discharge rates, which supports the rationale behind higher magnitude eccentric loading70,71. Furthermore, the acute improvements seen during the concentric phase after an accentuated eccentric contraction may be due to an enhanced neural drive72. This neuromuscular response may be explained in part due to enhanced cortical excitability, which seems to arise as a compensatory mechanism for spinal inhibition during eccentric action73.
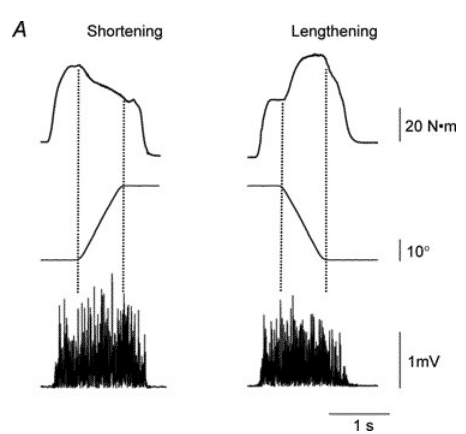
Figure 11 maximal shortening and lengthening contractions with the dorsiflexor muscles against a torque motor. Despite 10% less activation (EMG) during eccentric action, there was an increase of 15% in the torque produce during lenghtening69.
Moreover, It has been demonstrated that high-threshold motor units can be selectively recruited during eccentric contractions, particularly at fast eccentric velocities74. In addition to increased neural drive and selective recruitment of high threshold motor units, eccentric lengthening may lead to other recruitment strategies which may be related to changes in the motor-evoked potentials75 which may have the ability to create neural adaptations at the spinal level.76
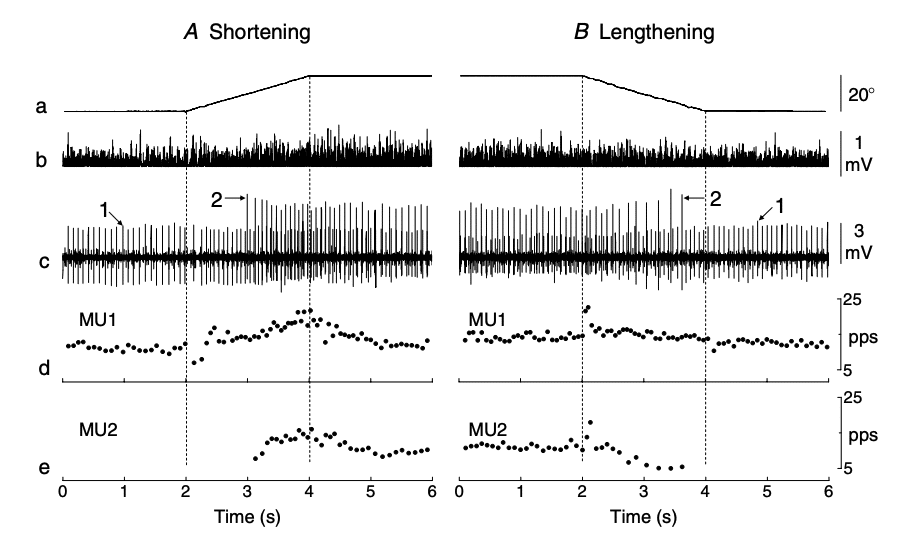
Figure 12 At the transition from the initial isometric contraction to the anisometric contraction, there was either a transient decrease (shortening contraction) or increase (lengthening contraction) in discharge rate due to an unloading reflex or stretch reflex, respectively69.
Bibliography
38.FRIDEN, J. & LIEBER, R. L. Structural and mechanical basis of exercise-induced muscle injury. Medicine & Science in Sports & Exercise 24, 521???530 (1992).
39.Friden, J. & Lieber, R. L. Eccentric exercise-induced injuries to contractile and cytoskeletal muscle fibre components. Acta Physiologica Scandinavica 171, 321–326 (2001).
40.Lauritzen, F., Paulsen, G., Raastad, T., Bergersen, L. H. & Owe, S. G. Gross ultrastructural changes and necrotic fiber segments in elbow flexor muscles after maximal voluntary eccentric action in humans. Journal of Applied Physiology 107, 1923–1934 (2009).
41.Hody, S., Croisier, J.-L., Bury, T., Rogister, B. & Leprince, P. Eccentric Muscle Contractions: Risks and Benefits. Frontiers in Physiology 10, (2019).
42.Takekura, H., Fujinami, N., Nishizawa, T., Ogasawara, H. & Kasuga, N. Eccentric exercise-induced morphological changes in the membrane systems involved in excitation-contraction coupling in rat skeletal muscle. The Journal of Physiology 533, 571–583 (2001).
43.Crameri, R. M. et al. Myofibre damage in human skeletal muscle: effects of electrical stimulationversusvoluntary contraction. The Journal of Physiology 583, 365–380 (2007).
44.Tidball, J. G. Mechanisms of Muscle Injury, Repair, and Regeneration. Comprehensive Physiology (2011) doi:10.1002/cphy.c100092.
45.Wang, Y. X. & Rudnicki, M. A. Satellite cells, the engines of muscle repair. Nature Reviews Molecular Cell Biology 13, 127–133 (2011).
46.Chen, B. & Shan, T. The role of satellite and other functional cell types in muscle repair and regeneration. Journal of Muscle Research and Cell Motility 40, 1–8 (2019).
47.Snijders, T. et al. A single bout of exercise activates skeletal muscle satellite cells during subsequent overnight recovery. Experimental Physiology 97, 762–773 (2012).
48.Fridén, J., Seger, J., Sjöström, M. & Ekblom, B. Adaptive Response in Human Skeletal Muscle Subjected to Prolonged Eccentric Training. International Journal of Sports Medicine 04, 177–183 (1983).
49.Clarkson, P. M. & Hubal, M. J. Exercise-Induced Muscle Damage in Humans. American Journal of Physical Medicine & Rehabilitation 81, S52–S69 (2002).
50.Chen, T. C. Variability in Muscle Damage After Eccentric Exercise and the Repeated Bout Effect. Research Quarterly for Exercise and Sport 77, 362–371 (2006).
51.McHugh, M. P. Recent advances in the understanding of the repeated bout effect: the protective effect against muscle damage from a single bout of eccentric exercise. Scandinavian Journal of Medicine and Science in Sports 13, 88–97 (2003).
52.NOSAKA, K. & CLARKSON, P. M. Muscle damage following repeated bouts of high force eccentric exercise. Medicine & Science in Sports & Exercise 27, 1263???1269 (1995).
53.HODY, S. et al. Human Muscle Proteome Modifications after Acute or Repeated Eccentric Exercises. Medicine & Science in Sports & Exercise 43, 2281–2296 (2011).
54.Nosaka, K., Newton, M. & Sacco, P. TIME COURSE OF ATTENUATION OF PROTECTIVE EFFECT AGAINST ECCENTRIC EXERCISE-INDUCED MSUCLE DAMAGE. Medicine & Science in Sports & Exercise 34, S184 (2002).
55.Nosaka, K., Newton, M. J. & Sacco, P. Attenuation of Protective Effect Against Eccentric Exercise-Induced Muscle Damage. Canadian Journal of Applied Physiology 30, 529–542 (2005).
56.Chen, T. C. et al. Low-intensity eccentric contractions attenuate muscle damage induced by subsequent maximal eccentric exercise of the knee extensors in the elderly. European Journal of Applied Physiology 113, 1005–1015 (2012).
57.McHugh, M. P., Connolly, D. A. J., Eston, R. G. & Gleim, G. W. Exercise-Induced Muscle Damage and Potential Mechanisms for the Repeated Bout Effect. Sports Medicine 27, 157–170 (1999).
58.Abbott, B. C., Bigland, B. & Ritchie, J. M. The physiological cost of negative work. The Journal of Physiology 117, 380–390 (1952).
59.LaStayo, P. C. et al. Eccentric Muscle Contractions: Their Contribution to Injury, Prevention, Rehabilitation, and Sport. Journal of Orthopaedic & Sports Physical Therapy 33, 557–571 (2003).
60.Franchi, M. V., Atherton, P. J., Maganaris, C. N. & Narici, M. V. Fascicle length does increase in response to longitudinal resistance training and in a contraction-mode specific manner. SpringerPlus 5, (2016).
61.Reeves, N. D., Maganaris, C. N., Longo, S. & Narici, M. V. Differential adaptations to eccentricversusconventional resistance training in older humans. Experimental Physiology 94, 825–833 (2009).
62.Narici, M., Franchi, M. & Maganaris, C. Muscle structural assembly and functional consequences. The Journal of Experimental Biology 219, 276–284 (2016).
63.MORGAN, D. L. & TALBOT, J. A. THE ADDITION OF SARCOMERES IN SERIES IS THE MAIN PROTECTIVE MECHANISM FOLLOWING ECCENTRIC EXERCISE. Journal of Mechanics in Medicine and Biology 02, 421–431 (2002).
64.TIMMINS, R. G. et al. Architectural Changes of the Biceps Femoris Long Head after Concentric or Eccentric Training. Medicine & Science in Sports & Exercise 48, 499–508 (2016).
65.Malliaras, P. et al. Patellar tendon adaptation in relation to load-intensity and contraction type. Journal of Biomechanics 46, 1893–1899 (2013).
66.Hyldahl, R. D. et al. Extracellular matrix remodeling and its contribution to protective adaptation following lengthening contractions in human muscle. The FASEB Journal 29, 2894–2904 (2015).
67.Brughelli, M. & Cronin, J. Altering the Length-Tension Relationship with Eccentric Exercise. Sports Medicine 37, 807–826 (2007).
68.Hoppeler, H. Moderate Load Eccentric Exercise; A Distinct Novel Training Modality. Frontiers in Physiology 7, (2016).
69.Duchateau, J. & Enoka, R. M. Neural control of shortening and lengthening contractions: influence of task constraints. The Journal of Physiology 586, 5853–5864 (2008).
70.TESCH, P. A., DUDLEY, G. A., DUVOISIN, M. R., HATHER, B. M. & HARRIS, R. T. Force and EMG signal patterns during repeated bouts of concentric or eccentric muscle actions. Acta Physiologica Scandinavica 138, 263–271 (1990).
71.Pasquet, B., Carpentier, A., Duchateau, J. & Hainaut, K. Muscle fatigue during concentric and eccentric contractions. Muscle & Nerve 23, 1727–1735 (2000).
72.Moore, C. A., Weiss, L. W., Schilling, B. K., Fry, A. C. & Li, Y. Acute Effects of Augmented Eccentric Loading on Jump Squat Performance. The Journal of Strength and Conditioning Research 21, 372 (2007).
73.Gruber, M., Linnamo, V., Strojnik, V., Rantalainen, T. & Avela, J. Excitability at the Motoneuron Pool and Motor Cortex Is Specifically Modulated in Lengthening Compared to Isometric Contractions. Journal of Neurophysiology 101, 2030–2040 (2009).
74.Nardone, A., Romanò, C. & Schieppati, M. Selective recruitment of high-threshold human motor units during voluntary isotonic lengthening of active muscles. The Journal of Physiology 409, 451–471 (1989).
75.Wagle, J. P. et al. Accentuated Eccentric Loading for Training and Performance: A Review. Sports Medicine 47, 2473–2495 (2017).
76.Lepley, L. K., Lepley, A. S., Onate, J. A. & Grooms, D. R. Eccentric Exercise to Enhance Neuromuscular Control. Sports Health: A Multidisciplinary Approach 9, 333–340 (2017).
